|
11/7/2023 0 Comments Caesium ion charge 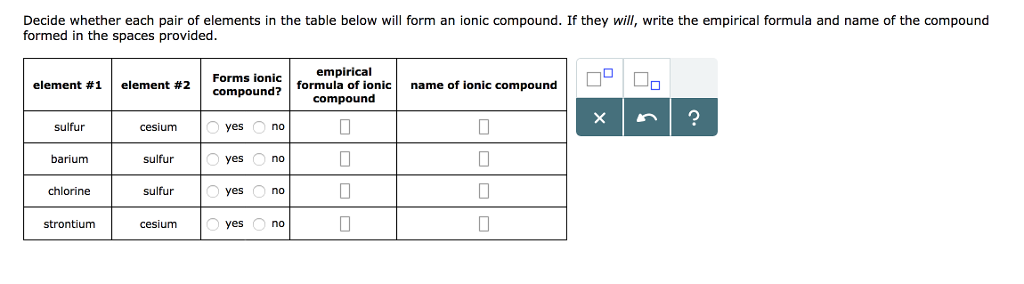
The chemical formula of calcium chloride can be given as CaCl 2. 
Therefore, the compound is electrically neutral. It is to make a note that the calcium cation holds a charge of +2 magnitude, and each chloride anion holds a charge of -1 magnitude. 
The calcium chloride structure molecules are represented below. The molecules of Calcium chloride feature two ionic bonds between a single calcium cation and two chloride anions. The important properties of calcium chloride can be tabulated as follows. Actually, it was considered a waste product until its uses were discovered. All of the entire early work was done with laboratory-prepared samples because it was not produced on a commercial scale until after the ammonia-soda process for the manufacture of soda ash was in operation. The Calcium chloride compound was discovered in the 15th century but received little study or attention until the latter part of the 18th century. This compound is widely used for de-icing and dust control.Ĭalcium Chloride can be prepared by the following:īy reacting hydrochloric and calcium carbonate solution acid orĭirectly from the limestone, a large amount is also produced as a by-product of the Solvay process. It has a very high enthalpy change of solution and is odourless. It is a crystalline solid white in colour at room temperature and is highly soluble in water, and hence it is hygroscopic in nature. We can also call it either Calcium dichloride or Calcium chloride anhydrous.Ĭalcium Chloride is an ionic compound composed of chlorine and calcium. CaCl 2 is said to be an ionic compound having a chemical name, Calcium Chloride. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
